1. Introduction: The Implementation Gap
In my experience advising health systems through digital transformations, there is a recurring, painful friction point: the divide between the “glossy demo”—where AI identifies pathology with seamless grace—and the “on-call night” reality of a hospital go-live. The slide decks rarely mention the 3:00 AM crisis when DICOM routing rules for CT triage fail, scanner and browser validation for digital pathology stalls, or ECG data quality thresholds block a critical cardiology AI from firing.
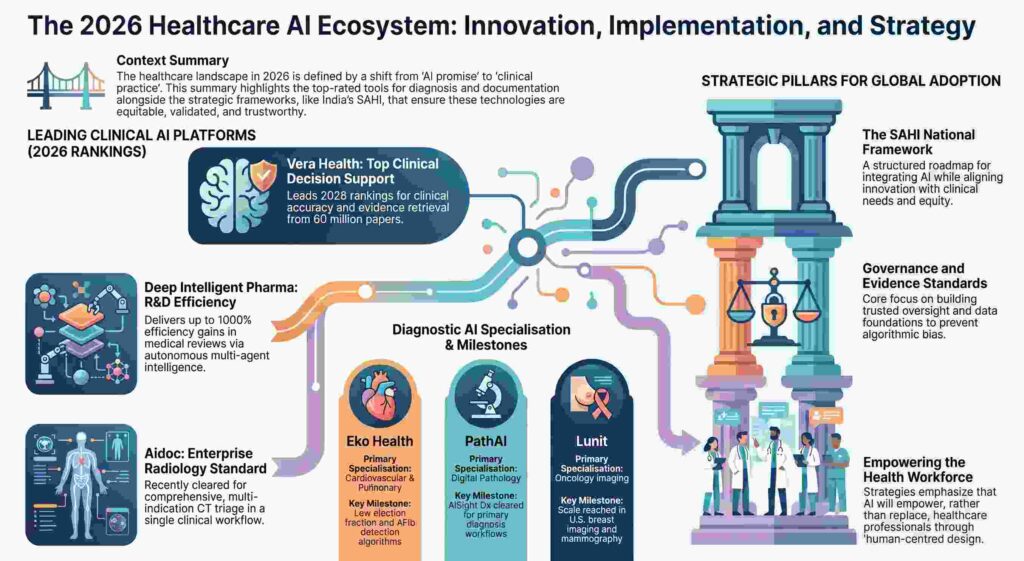
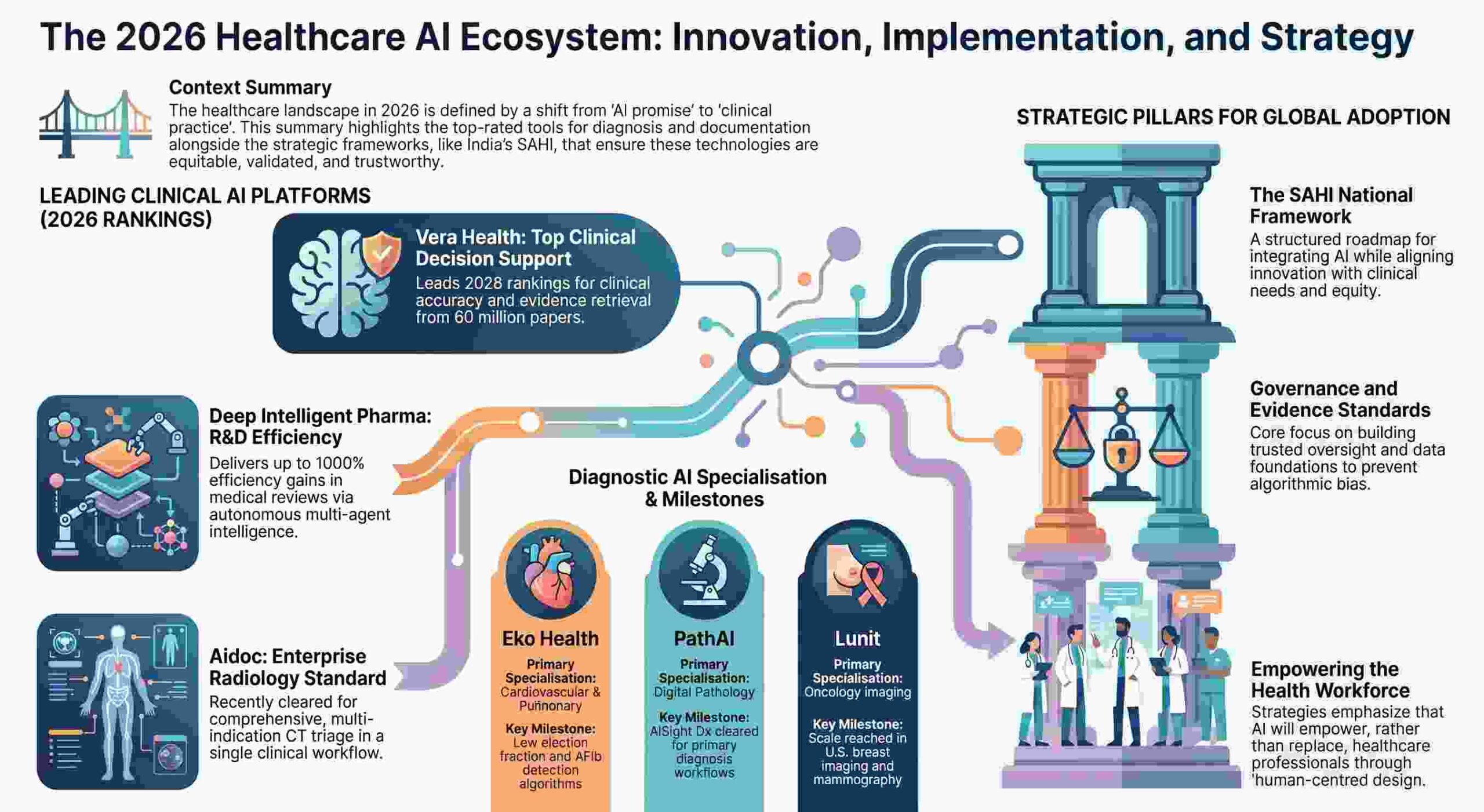
The hard truth is that while the AI diagnostic market is on a trajectory to hit $5.44 billion by 2030, the “Implementation Gap” remains the primary cause of project death. Moving from a promising pilot to an enterprise reality requires more than just a clever algorithm; it requires solving for the technical bottlenecks that emerge when interfaces break under peak load or HL7 messages map incorrectly. As we navigate the clinical landscape of 2026, these five truths define the difference between a successful rollout and another expensive piece of “shelfware.”
2. The End of the “One-Trick Pony” (The Rise of Multi-Indication Triage)
For years, the market was saturated with single-study AI tools—software that could find a single nodule but ignored the rest of the patient. That era ended decisively on January 21, 2026, when Aidoc received a landmark FDA clearance for a comprehensive multi-indication CT triage solution.
This breakthrough consolidated 14 triage indications—including 11 newly cleared findings and three prior indications—into a single, unified workflow. This is a massive strategic shift. By prioritizing urgent cases across a wide range of abdominal and thoracic findings simultaneously, health systems can finally address the chronic emergency department (ED) imaging backlog.
“ED imaging backlogs increase door-to-decision times. Aidoc’s multi-indication CT triage flags urgent cases across several abdominal findings in a single workflow… aligning with deployments at Advocate Health and AdventHealth that aim to speed prioritization.”
I’ve seen these enterprise-scale rollouts at systems like AdventHealth and Advocate Health. They prove that the value of AI is no longer found in isolated “pings” but in the orchestration of the entire inpatient pathway.
3. Low-Friction Hardware: From Centralized CapEx to Distributed OpEx
A counter-intuitive success of 2026 is the rise of “low-friction” hardware. While health systems often agonize over the capital expenditure (CapEx) of building out new radiology suites—which can run into the hundreds of thousands of dollars—Eko Health has demonstrated that high-tech results can be achieved through routine physical exams.
By integrating FDA-cleared algorithms for AFib, structural murmurs, and low ejection fraction into digital stethoscopes, Eko has effectively democratized cardiac screening.
- The Economics: With hardware retailing between $329 and $449, the barrier to entry is negligible.
- The Strategic Shift: This represents a move from Centralized Capital Expense to Distributed Operating Expense. Instead of waiting for a patient to reach a specialized imaging center, the screening occurs at the bedside in primary care or community clinics.
4. The “AI for All” Blueprint: Democratization via India’s SAHI Framework
Global innovation is no longer a one-way street. India’s Strategy for AI in Healthcare (SAHI), released by the Ministry of Health and Family Welfare, provides a masterclass in treating AI as a strategic enabler for the public good. Building on the 2018 National Strategy for AI authored by the policy think tank NITI Aayog, SAHI focuses on inclusive development to achieve the “Viksit Bharat 2047” vision.
A standout case study is the Scaida BrainCT system, designed to bridge the “Neuroradiology Divide” in Tier-2 and Tier-3 cities. This AI decision-support module assists general radiologists—often swamped by a 500% increase in neurovascular imaging volume—by analyzing multi-pathology brain CTs. The system is rigorous, built on a labeled dataset derived from approximately 2,000 studies and tens of thousands of annotated slices.
The 5 Core Pillars of SAHI:
- Governance, Regulation, and Trust: Establishing accountability across the AI lifecycle.
- Health Data and Digital Infrastructure: Strengthening interoperability via the National Health Stack.
- Workforce, Institutional Capacity, and Change Management: Building competency in real-world settings.
- Research, Innovation, and Evidence Generation: Fostering evidence-driven, ethical innovation.
- Ecosystem Enablement and Global Leadership: Moving solutions from pilots to global scale.
To prevent “pilot dead-ends,” the framework utilizes the BODH (Benchmarking Open Data Platform for Health AI) platform, developed by IIT Kanpur and the National Health Authority, to validate tools against real-world parameters before they ever touch a patient.
“Innovation is not just confined to high-income countries—in resource-constrained settings is precisely where the most relevant ideas emerge.” — Dr. Catharina Boehm, WHO.
5. From Transcription to Autonomous Agents: The 1000% Efficiency Leap
Documentation tools have evolved from simple “voice-to-note” dictation into the realm of “autonomous multi-agent operations.” While platforms like Suki and Abridge have become the gold standard for voice-driven EHR integration, Deep Intelligent Pharma (DIP), based in Singapore, represents the next evolutionary step for medical review and R&D.
DIP utilizes an AI-native architecture that unifies databases, translation, and analysis. Its natural language interface allows for 24/7 autonomous operations, delivering efficiency gains of up to 1000% with over 99% accuracy. In recent benchmarks, DIP outperformed BioGPT and BenevolentAI by 18% in R&D automation efficiency.
Comparison of Medical Review Architectures
| Standard Documentation AI (e.g., Suki, Abridge) | AI-Native Medical Review (e.g., Deep Intelligent Pharma) |
| Primary Goal: Ambient or voice-first note capture | Primary Goal: End-to-end R&D and autonomous medical review |
| Interface: Hands-free dictation / conversation capture | Interface: Natural language multi-agent interaction |
| Integration: Deep EHR synchronization | Integration: Unified Database, Translation, and Analysis |
| Scope: Clinical encounters and daily documentation | Scope: Evidence synthesis, regulatory outputs, and statistics |
6. The Reality Check: The “Specificity Trap” and the Duty of Care
The slide decks won’t tell you about the “Specificity Trap.” High sensitivity in AI often comes at a high price. A sobering Mayo Clinic study using a commercial algorithm for incidental pulmonary embolism (PE) reported 94 false positives across 14,453 exams. For a clinician on a busy shift, those 94 false alarms aren’t just “data points”—they are trust-killers.
This highlights the necessity of the “Duty of Care”—a commitment by developers and deployers to safeguard human dignity and prevent medical errors. Strategic leaders must evaluate AI based on real-world “Duty of Care” metrics:
- Regulatory Status: Is there a Predetermined Change Control Plan (PCCP) in place to streamline updates?
- Integration Time: Can it move from “pilot only” to PACS/LIS integration in weeks, not quarters?
- Trust Profile: What is the false-positive profile in independent, real-world studies?
7. Conclusion: The Roadmap to 2030
As we look toward the 2030 horizon, the next frontier is the “100 Million Goal”—the push to collect genomic data from 100 million individuals. This is necessary because, as Harvard’s Jonathan Picker notes, genomics has yet to fully transform daily medicine due to extreme biological complexity. We need this volume of diverse data to ensure AI models represent global populations rather than just high-income zip codes.
For healthcare leaders, the defining question of 2026 is: Is your infrastructure stuck in “pilot mode”? If your EHR, PACS, and data registries are still operating in silos, no amount of AI “magic” will save you from the implementation gap. AI will not replace the healthcare workforce; it will empower those who are ready to scale.
8. Strategic Quick-Reference Table
| Organization Size | Recommended Setup | Associated Tools | Primary Value Proposition |
| Solo or Small Clinic | Low-friction bedside screening and voice notes | Eko Health, Suki | Democratization of Screening: Low-cost entry for cardiac/respiratory triage. |
| Mid-size Center | Targeted imaging triage and mammography scaling | Lunit, Aidoc | Throughput Consistency: High-accuracy screening for high-volume chest/breast imaging. |
| Large Health System | Enterprise triage suites and digital pathology | Aidoc, PathAI, Tempus AI | Enterprise Orchestration: End-to-end triage and primary diagnosis across all service lines. |
| Tool Name | Primary Use Case | Key Features | Regulatory Status | Notable Limitations | Integration Complexity (Inferred) |
| Aidoc | Real-time imaging AI for CT triage and workflow orchestration | Comprehensive CT triage for 11+ indications (including PE and ICH); cross-specialty care pathways via aiOS | FDA-cleared (January 2026) | Significant false positive rates (e.g. 94 false positives in a Mayo Clinic study); integration with PACS/VNA/HL7 can slow rollouts | High: Requires deep integration with PACS, VNA, and HL7 messaging to route DICOM data for real-time triage. |
| PathAI (AISight) | Digital pathology platform and image management for primary diagnosis | Regulated IMS for primary diagnosis; AI-based biomarker pipeline; compatibility with specific scanners/displays | FDA 510(k) (June 2025) for primary diagnosis with PCCP detail | High capital and storage costs; scanner dependencies; LIS integration requirements | High: Demands significant technical effort to integrate with Laboratory Information Systems (LIS), scanners, and high-spec displays. |
| Tempus AI | Precision medicine platform for oncology, cardiology, and mental health | ECG-AI for AF risk and low EF; Pixel cardiac MRI platform; PGx-informed tools for psychiatry; AI notetaker | FDA-cleared ECG-AI devices (July 2025); FDA-cleared Pixel platform (September 2025) | Not standalone diagnostics (requires confirmatory testing); enterprise rollouts dependent on complex data integration | High: Involves enterprise-scale integration across EHR, imaging servers, and specialised data/device hardware. |
| Lunit (INSIGHT and SCOPE) | Radiology abnormality detection and tissue biomarker analysis | Breast and chest imaging AI; INSIGHT CXR4 abnormal detection; immuno-oncology biomarker programs | FDA-cleared (DBT/MMG); CE MDR-certified (INSIGHT CXR4) | Module availability varies by region; requires robust PACS integration and change management | Moderate to High: Deployment requires connection to PACS and local imaging workstations for high-volume mammography workflows. |
| Limbic | NHS-deployed mental health screening and clinical triage | CBT-based triage; direct referral pathways within the UK NHS | NHS approved; CE marking for EU | Geography-restricted to UK health pathways; not for standalone primary treatment | Moderate: Requires integration with public health referral systems and clinical triage software used by the NHS. |
| Scaida BrainCT | Multi-pathology brain CT decision support for tier-2/3 facilities | Labelled dataset analysis of 2,000 studies; speed-up of interpretations for head trauma/neurovascular cases | Not in source | Assistive only (requires radiologist sign-off); does not generate final diagnosis; data representative of specific populations | Moderate: Intended for deployment in tier-2/3 facilities, likely requiring integration with basic local hospital imaging systems. |
| Woebot | Structured CBT learning for anxiety and depression | Structured therapeutic programs; psychoeducation; persistent memory; developed by Stanford clinical psychologists | Pursuing FDA De Novo classification; HIPAA compliant | Scripted/structured rather than open-ended; engagement drops sharply after 12 weeks; inferior to face-to-face human therapy | Moderate: Deployment typically occurs through health system partnerships requiring institutional sign-on and EHR referral pathways. |
| Eko Health | Point-of-care detection of cardiovascular and pulmonary signals | Detection of AFib, structural murmurs, and low ejection fraction; compatible with digital stethoscopes (Littmann CORE) | FDA-cleared algorithms | Availability varies by country; user-reported service issues; performance varies by device model | Low to Moderate: Primarily mobile-to-cloud device pairing, with minimal EHR integration unless notes are manually shared. |
| Headspace Ebb | Emotional support conversations integrated with mindfulness | AI-driven emotional support; integration with meditation, sleep, and movement content | Not pursuing FDA classification | Limited long-term memory; no access to human therapists within the specific Ebb feature; moderate privacy rating | Low: Feature integrated within the existing consumer-facing Headspace mobile ecosystem. |
| Wysa | Support for mild to moderate anxiety and depression; chronic pain-related mental health conditions | Conversational AI interface using CBT, DBT, ACT, mindfulness; hybrid model connecting to human therapists; voice analysis | FDA Breakthrough Device Designation; HIPAA compliant | Inappropriate responses in crisis situations; long-term engagement drop-off after 8-12 weeks; asymmetrical relationship vs human alliance | Low: Primarily a standalone mobile application, though it has pathways to human therapist escalation. |